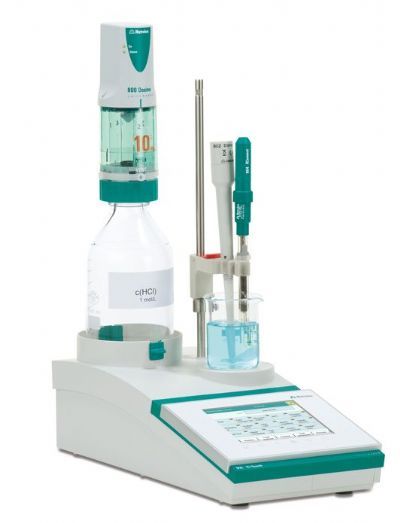
化学药中主要物质含量分析检测方案(制备液相色谱)
本论文以两种海洋真菌草酸青霉菌和构巢曲菌为研究对象,对其代谢产物用Biotage isolera进行了分离纯化,同时进行了结构解析以及立体构型的鉴定。一共分离鉴定了31种化合物,其中1个新化合物以及3个化合物该种属内首次发现。耐士科技作为Biotage中国区总代理,以最优质的服务提供Biotage全系产品。Biotage Isolera 系列是世界上最智能的快速纯化系统,它拥有自己独创的智能参数设置,共有3个系统,多种配置可选。可以让化学家们轻松地完成对从mg级到150g以上样品 的更好的分离。
创新的TLC-to-gradient专利技术可以根据薄层层析色谱的数据自动产生适合样品的溶剂洗脱梯度,并建议适合该样品量的色谱柱。
通过双波长检测收集馏分,最多可以在单一梯度下同时使用四种溶剂进行洗脱,以达到最大限度提高纯度和收率的目的。
通过梯度优化功能,可以实现加大上样量同 时减少溶剂使用量。
检测样品:
化药新药研发
检测项:
临床前研究
上海鑫欣生物科技有限公司
查看联系电话
前往展位
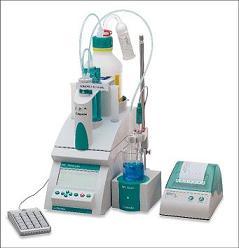
化学药中主要物质含量分析检测方案(制备液相色谱)
Clandestine laboratories constantly produce new synthetic cannabinoids to circumvent legislative efforts,complicating toxicological analysis. No extensive synthetic cannabinoid quantitative urinary methodsare reported in the literature. We developed and validated a liquid chromatography–tandem mass spec-trometric (LC–MS/MS) method for simultaneously quantifying JWH-018, JWH-019, JWH-073, JWH-081,JWH-122, JWH 200, JWH-210, JWH-250, JWH-398, RCS-4, AM-2201, MAM-2201, UR-144, CP 47,497-C7,CP 47,497-C8 and their metabolites, and JWH-203, AM-694, RCS-8, XLR-11 and HU-210 parent com-pounds in urine. Non-chromatographically resolved alkyl hydroxy metabolite isomers were consideredsemi-quantitative. Glucuronidase hydrolyzed urine was extracted with 1 ml Biotage SLE+ columns.Specimens were reconstituted in 150 L mobile phase consisting of 50% A (0.01% formic acid in water)and 50% B (0.01% formic acid in 50:50 methanol:acetonitrile). 4 and 25 L injections were performed toacquire data in positive and negative ionization modes, respectively. The LC–MS/MS instrument consistedof a Shimadzu UFLCxr system and an ABSciex 5500 Qtrap massspectrometer with an electrospray source.Gradient chromatographic separation was achieved utilizing a Restek Ultra Biphenyl column with a0.5 ml/min flow rate and an overall run time of 19.5 and 11.4 min for positive and negative mode methods,respectively. Quantification was by multiple reaction monitoring with CP 47,497 compounds and HU-210ionized via negative polarity; all other analytes were acquired in positive mode. Lower and upper limitsof linearity were 0.1–1.0 and 50–100 g/l (r2> 0.994).
检测样品:
化药新药研发
检测项:
临床前研究
上海鑫欣生物科技有限公司
查看联系电话
前往展位
化学药中有效成分含量分析检测方案
Discovery DSC-18Lt固相萃取和Discovery C18液相色谱分析血清中巴比妥酸盐,回收率都能达到95%以上。
关于Supelco
美国Supelco公司成立于1966年,一直致力于色谱耗材的研究和生产,是色谱耗材的专业生产公司。超过40年在色谱和分析领域的技术经验,拥有多项专利技术,提供范围广泛的产品:气相色谱柱(包括手性柱)和配件、液相色谱柱(包括手性柱)和配件、固相萃取小柱和装置、固相微萃取手柄和萃取头、空气检测产品、分析标准品和样品瓶等。1993年,Supelco正式加入美国Sigma-Aldrich公司,成为Sigma-Aldrich公司旗下分析业务的专业品牌。
检测样品:
化药新药研发
检测项:
临床前研究
默克化工技术(上海)有限公司
查看联系电话
前往展位
化学药中有效成分含量分析检测方案
Discovery DSC-8 固相萃取和Discovery C18液相色谱分析血清中安定和代谢物。回收率都在90%以上。
关于Supelco
美国Supelco公司成立于1966年,一直致力于色谱耗材的研究和生产,是色谱耗材的专业生产公司。超过40年在色谱和分析领域的技术经验,拥有多项专利技术,提供范围广泛的产品:气相色谱柱(包括手性柱)和配件、液相色谱柱(包括手性柱)和配件、固相萃取小柱和装置、固相微萃取手柄和萃取头、空气检测产品、分析标准品和样品瓶等。1993年,Supelco正式加入美国Sigma-Aldrich公司,成为Sigma-Aldrich公司旗下分析业务的专业品牌。
检测样品:
化药新药研发
检测项:
临床前研究
默克化工技术(上海)有限公司
查看联系电话
前往展位
化学药中有效成分含量分析检测方案
代谢物解析测定生物体内小分子代谢物以把握其变化,广泛使用具有高色谱分离能力、高灵敏度且可稳定测定的GC-MS。但在分析多基质的生物样品时,使用GC-MS时,目标成分有时难以与基质分离。 此次岛津推出了基于具备出色MS分离能力的岛津GC-MS/MS的鼠尿中代谢物的分析方案,并与基于GC-MS的分析数据进行了比较。GC-MS模式的SIM测定时,因基质的影响,难以进行Glycerol-3TMS、Suberic acid-2TMS的化合物鉴定、定量。而GC-MS/MS的MRM测定将基质影响质量分离,可以进行鉴定・定量。GC-MS/MS可以有效地分析存在大量杂质的生物样品。
检测样品:
化药新药研发
检测项:
临床前研究
岛津企业管理(中国)有限公司
查看联系电话
前往展位
利培酮的活性对映体代谢物中分离代谢稳定性检测方案(液相色谱仪)
许多候选药物及其代谢物都含有一个或多个手性中心。识别和监测可能存在的各种对映体是药物开发过程中至关重要的一步。众所周知,超临界流体色谱(SFC)是一种可进行手性分离的高效技术。在本技术简报中,我们展示了利用UPC2/MS/MS对利培酮的活性代谢物9-羟基利培酮进行对映体分离和检测。研究人员使用9-羟基利培酮标准品进行方法优化,然后将其应用于在人肝脏微粒体中温育的利培酮样品。实验监测了从母体化合物到羟基代谢物的转化过程。
检测样品:
化药新药研发
检测项:
临床前研究
沃特世科技(上海)有限公司(Waters)
查看联系电话
前往展位
头孢噻肟钠与氧氟沙星注射液中配伍的稳定性研究检测方案
摘要 目的 研究头孢噻肟钠与氧氟沙星注射液配伍稳定性。 方法 建立头孢噻肟钠与氧氟沙星混合液紫外分光光度法含量测定方法,分析混合液在24℃下8h内两药含量,并观察其外观、pH值及紫外光谱。结果 4h内混合液外观、pH值及紫外光谱无明显变化,6h后两药含量测定结果较4h偏高,8h后混合液紫外光谱发生改变。结论 在24℃下,头孢噻肟钠与氧氟沙星注射液配伍后4h内稳定性较好,临床上两药配伍使用应在4h内完成。
关键词 头孢噻肟钠;氧氟沙星;配伍稳定性
检测样品:
化药新药研发
检测项:
临床前研究
北京普析通用仪器有限责任公司
查看联系电话
前往展位
基于1T小动物磁共振成像小鼠生理特征定量计算
1.0T小动物核磁共振成像仪是纽迈2016年推出的新品,是目前纽迈分析磁场强度最高的核磁共振成像仪。1.0T的永磁体,优质的磁场均匀性,搭载纽迈高性能梯度系统,提供更高的图像分辨率,为科研提供更多的研究方向和思路;此外,根据不同动物尺寸大小量身设计匹配线圈,精准调谐,最大限度提高信噪比和图像清晰度,并可进行薄层(低至0.8mm)任意角度任意层面扫描。该仪器保留了纽迈经典的三步法成像软件,搭配新开发的多功能核磁共振图像处理软件,让后续图像的处理简便而高效。
检测样品:
化药新药研发
检测项:
临床前研究
苏州纽迈分析仪器股份有限公司
查看联系电话
前往展位
化学药新药研发中人血清蛋白与干扰素α-2b融合蛋白检测方案(毛细管电泳仪)
Capel 205高效毛细管电泳仪是一种快速、简便的分析仪器,可应用于多个行业,进行定性和定量分析;仪器配备液体恒温系统,提高了检测的稳定性;配备自动进样器可实现完全自动进样和分析;可通过电脑实现仪器的控制。成熟的仪器、优化的配置、大量的应用方法包集一体,被用户称为目前性价比最优的毛细管电泳仪、产品方法符合EPA 6500,水中阴离子检测;ASTM D 6508-00、ASTM D7881、ASTM D7882,GB/T 30921.1-2014 、OVI(国际葡萄和葡萄酒组织)等方法标准。
检测样品:
化药新药研发
检测项:
临床前研究
LUMEX INSTRUMENTS (鲁美科思分析仪器)
查看联系电话
前往展位
新品药物中研发、制备及质控过程检测方案(挤出机)
药物热熔挤出(HME)技术作为一种新型的药物传递技术,创造性地将加工技术与药学结合起来进行药物传递研究,专为提高难溶性 APIs的溶解度和生物利用度,研发新型缓控释制剂,制备掩味微丸或者其它特殊形状的制剂,例如植入剂等,应用前景广阔。其结合了在固体分散技术和机械制备的诸多优势,实现了无粉尘、连续化操作、良好的重现性,以及极高的生产效率。
Thermo ScientificTM Pharma 双螺杆挤出机系列在本质上属于连续过程仪器,支持现代制剂科学采用四种工艺模式创造新型固体药物剂型 : 药物热熔挤出、熔融制粒、干法制粒、湿法制粒、和湿法挤出,创造新型固体药物剂型,且可通过一台设备实现五种工艺模式交替使用。通过与近红外(NIR)光谱联用实时同步检测 APIs和辅料浓度,作为过程分析技术(PAT)和质量源于设计原则(Qbd)工具,加深理解工艺过程和即时过程控制。
检测样品:
化药新药研发
检测项:
临床前研究
赛默飞世尔科技材料表征
查看联系电话
前往展位
仪器信息网行业应用栏目为您提供31篇化药新药研发检测方案,可分别用于含量测定检测、限度检查检测、注射剂及特殊剂型相关检测、组学研究检测、化合物发现检测、临床前研究检测、分子量检测,参考标准主要有等