方案详情
文
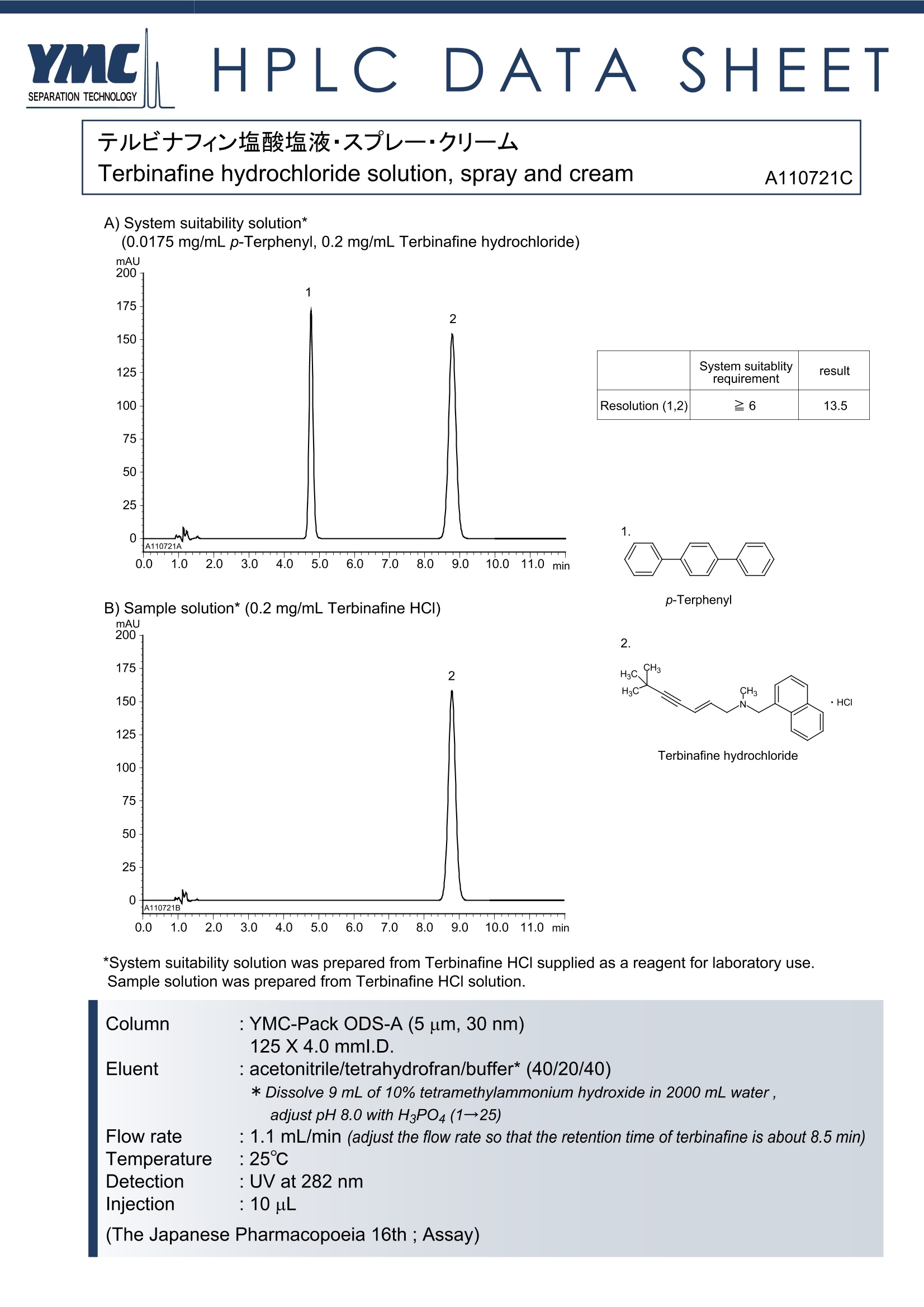
按日本药典规定的液相色谱条件,用YMC-Pack ODS-A色谱柱(规格:5um,125*4.0mm,30nm,货号:AA30S05-R504WT)测定盐酸特比萘芬溶液、喷雾剂、乳膏剂中盐酸特比萘芬的含量,分离度等完全符合药典要求,测定结果令人满意。
方案详情

HPLCDATA'SHEET 儿十7酸液液·又一·一Terbinafine hydrochloride solution, spray and cream A110721C A) System suitability solution*(0.0175 mg/mL p-Terphenyl, 0.2 mg/mL Terbinafine hydrochloride)mAU 200 System suitablity result requirement Resolution (1,2) 26 13.5 1. B) Sample solution* (0.2 mg/mL Terbinafine HCI) p-Terphenyl mAU 2. CH3 H3C CH3 ·HC Terbinafine hydrochloride *System suitability solution was prepared from Terbinafine HCI supplied as a reagent for laboratory use.Sample solution was prepared from Terbinafine HCI solution. Column : YMC-Pack ODS-A (5 um, 30 nm) 125X4.0mml.D. Eluent : acetonitrile/tetrahydrofran/buffer* (40/20/40) * Dissolve 9 mL of 10% tetramethylammonium hydroxide in 2000 mL water, adjust pH 8.0 with H3PO4(1→25) Flow rate : 1.1 mL/min (adjust the flow rate so that the retention time of terbinafine is about 8.5 min) Temperature :25℃ Detection : UV at 282 nm Injection : 10uL (The Japanese Pharmacopoeia 16th ; Assay) 盐酸特比萘芬乳膏(喷雾剂、溶液)含量测定色谱条件(JP VXI) 仪 器:液相色谱仪检测器:UV (282nm)色谱柱:YMC-Pack ODS-A色谱柱(内径:4.0mm 长度:125mm 粒径:3um 孔径:30nm)流动相:乙腈-四氢呋喃-缓冲溶液* 2:1:2 *缓冲溶液:将9克四甲基氢氧化铵溶解于2000ml水中,并用稀磷酸溶液(1:25)调pH至8.0柱 温: 25℃流 速:1.1ml/min(调整特比萘芬色谱峰保留时间约8.5分钟)进样量:10ul(0.2mg/ml) 系统分离度:取适量盐酸特比萘芬和p-三联苯溶于甲醇,浓度分别为0.2mg/ml和0.0175mg/ml。取10ul进样,p-三联苯和特比萘芬的分离度不小于6。 系统重复性:RSD≤1.0%
确定
还剩1页未读,是否继续阅读?
深圳凯米斯科技有限公司为您提供《盐酸特比萘芬乳膏,盐酸特比萘芬溶液,盐酸特比萘芬喷雾剂中主要物质含量检测方案(液相色谱柱)》,该方案主要用于化药制剂中含量测定检测,参考标准--,《盐酸特比萘芬乳膏,盐酸特比萘芬溶液,盐酸特比萘芬喷雾剂中主要物质含量检测方案(液相色谱柱)》用到的仪器有
相关方案
更多









