Quality assurance of lithium-ion battery precursor chemicals by Agilent 5800 VDV ICP-OES
方案详情

Driven by demand from high tech industries for lithium-ion batteries (LIBs), the global market for lithium (Li) compounds is expected to reach USD 26.7 billion by 2030 (1). Commercial production of Li compounds relies on extraction of Li from two naturally occurring sources; brine, principally in South America, and hard rock minerals such as spodumene, from orebodies found mainly in Australia and China. Traditionally, lithium hydroxide (LiOH) was converted from brine-extracted lithium carbonate (Li2CO3). However, recent advances in technology (e.g., membrane technology) have enabled the production of LiOH directly from brine. Both Li2CO3 andLiOH can be produced directly from hard rock minerals.
确定


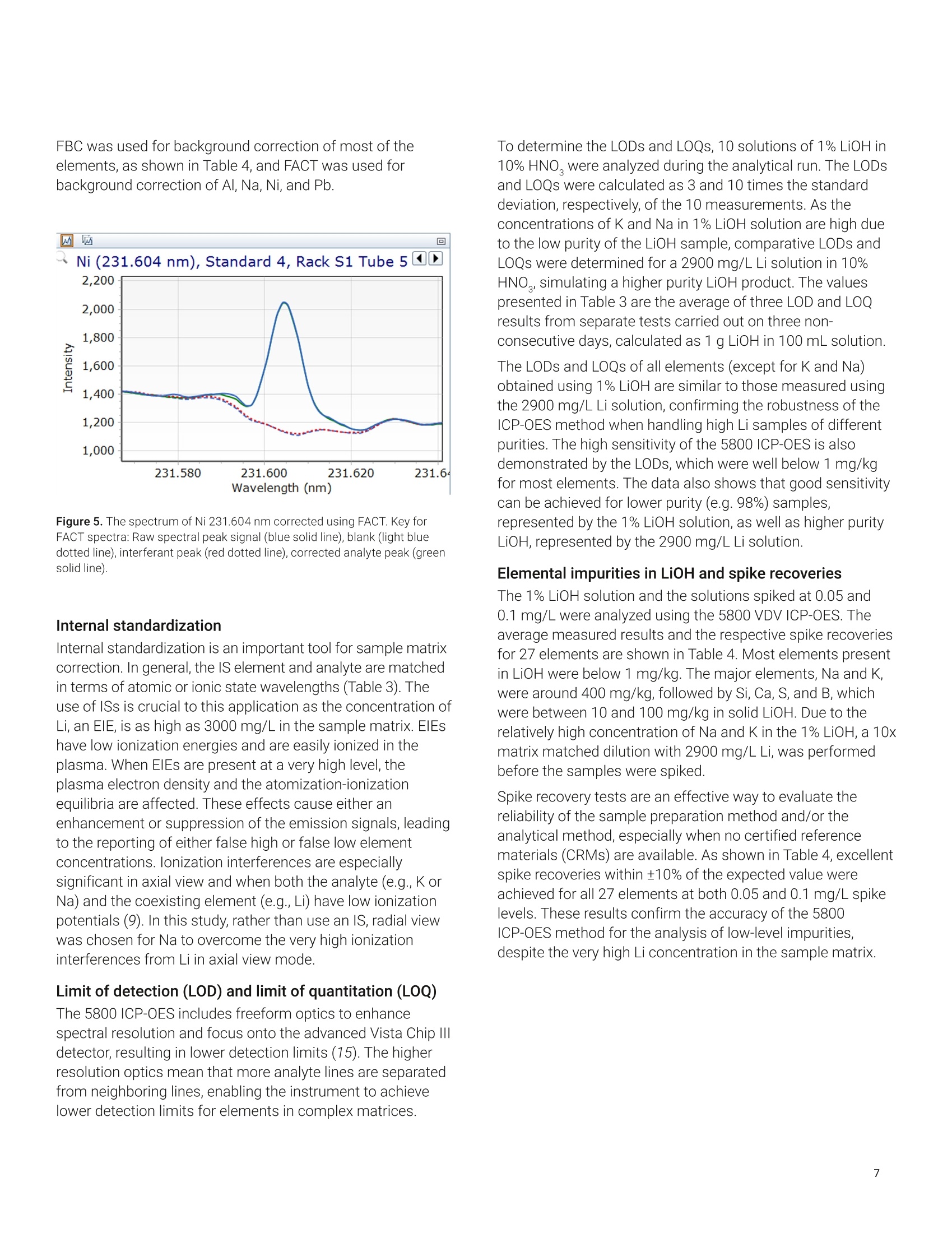
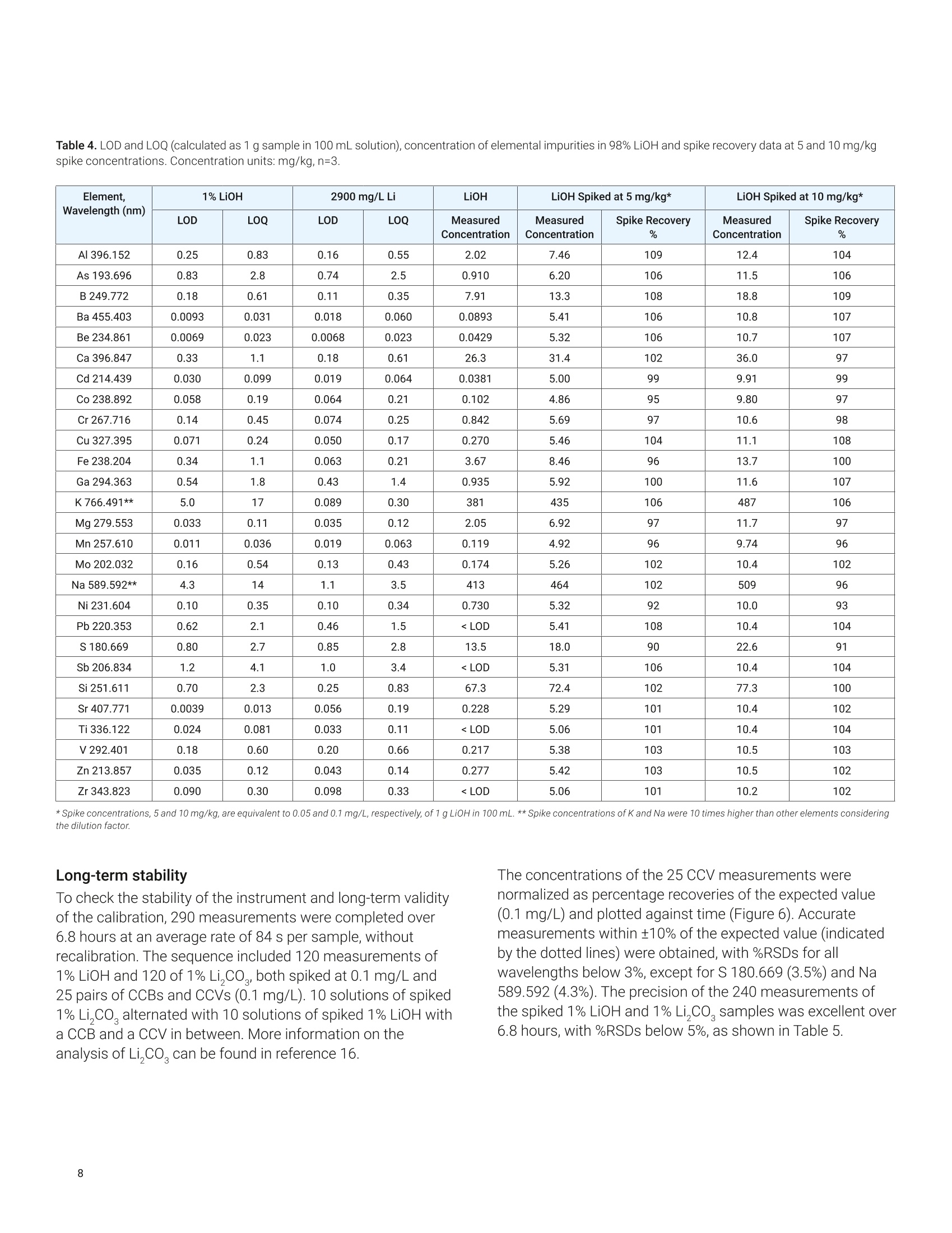
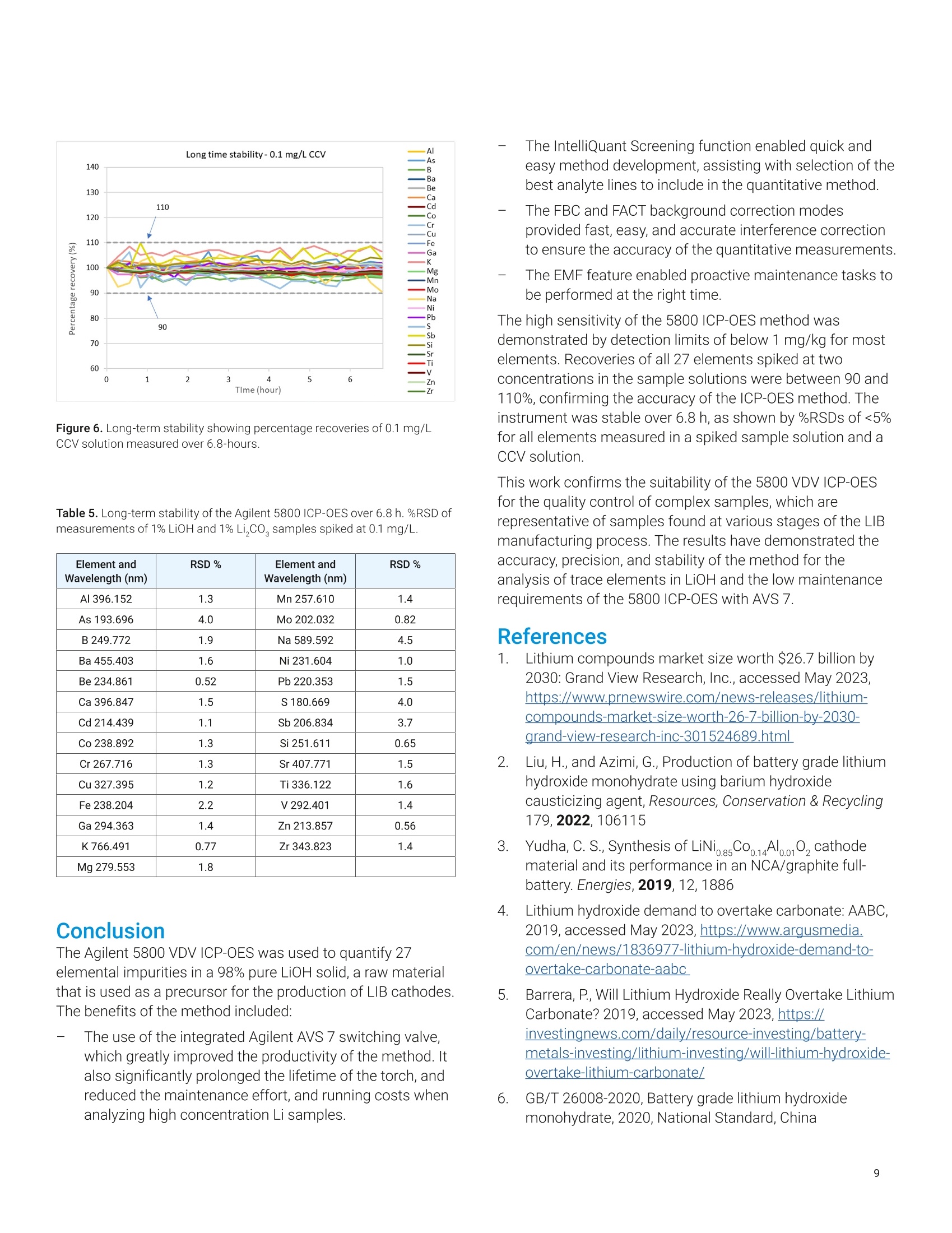

还剩9页未读,是否继续阅读?
安捷伦科技(中国)有限公司为您提供《Determination of Elemental Impurities in Lithium Hydroxide Using ICP-OES》,该方案主要用于锂电池中理化性能检测,参考标准--,《Determination of Elemental Impurities in Lithium Hydroxide Using ICP-OES》用到的仪器有Agilent 5800 ICP-OES
推荐专场
相关方案
更多
该厂商其他方案
更多
















