推荐厂家
暂无
暂无



 400-831-3689
400-831-3689
 留言咨询
留言咨询

 400-805-8969
400-805-8969
 留言咨询
留言咨询

 400-805-8969
400-805-8969
 留言咨询
留言咨询



[align=left][font='times new roman'][size=18px]S[/size][/font][font='times new roman'][size=18px]il[/size][/font][font='times new roman'][size=18px]-P-C12[/size][/font][font='times new roman'][size=18px]色谱柱分离硫脲类化合物[/size][/font][font='times new roman'][size=18px]/[/size][/font][font='times new roman'][size=18px]苯酚类化合物[/size][/font][font='times new roman'][size=18px]/[/size][/font][font='times new roman'][size=18px]磺胺类化合物[/size][/font][/align][font='times new roman'][size=16px]为了考察[/size][/font][font='times new roman'][size=16px]Sil[/size][/font][font='times new roman'][size=16px]-P-C12[/size][/font][font='times new roman'][size=16px]色谱柱的分离性能,选取了七种小分子化合物[/size][/font][font='times new roman'][size=16px]——[/size][/font][font='times new roman'][size=16px]硫脲类、苯酚类、磺胺类、酰胺类、黄酮类、核苷[/size][/font][font='times new roman'][size=16px]/[/size][/font][font='times new roman'][size=16px]核酸碱基类和烷基苯类。首先是三种硫脲类化合物[/size][/font][font='times new roman'][size=16px],[/size][/font][font='times new roman'][size=16px]图[/size][/font][font='times new roman'][size=16px]1[/size][/font][font='times new roman'][size=16px]为[/size][/font][font='times new roman'][size=16px]二乙基硫脲、亚乙基硫脲和烯丙基硫脲[/size][/font][font='times new roman'][size=16px]的色谱分离图。以乙腈[/size][/font][font='times new roman'][size=16px]-[/size][/font][font='times new roman'][size=16px]水([/size][/font][font='times new roman'][size=16px]97[/size][/font][font='times new roman'][size=16px]:[/size][/font][font='times new roman'][size=16px]3[/size][/font][font='times new roman'][size=16px],[/size][/font][font='times new roman'][size=16px]v/v[/size][/font][font='times new roman'][size=16px])进行洗脱,三种物质可在[/size][/font][font='times new roman'][size=16px]6 min[/size][/font][font='times new roman'][size=16px]内达到基线分离,分离度分别为[/size][/font][font='times new roman'][size=16px]9.21[/size][/font][font='times new roman'][size=16px]和[/size][/font][font='times new roman'][size=16px]5.04[/size][/font][font='times new roman'][size=16px],拖尾因子[/size][/font][font='times new roman'][size=16px]分别为[/size][/font][font='times new roman'][size=16px]1[/size][/font][font='times new roman'][size=16px].07[/size][/font][font='times new roman'][size=16px]、[/size][/font][font='times new roman'][size=16px]1[/size][/font][font='times new roman'][size=16px].45[/size][/font][font='times new roman'][size=16px]和[/size][/font][font='times new roman'][size=16px]1[/size][/font][font='times new roman'][size=16px].22[/size][/font][font='times new roman'][size=16px],最高理论塔板数为[/size][/font][font='times new roman'][size=16px]6[/size][/font][font='times new roman'][size=16px]1[/size][/font][font='times new roman'][size=16px],[/size][/font][font='times new roman'][size=16px]1[/size][/font][font='times new roman'][size=16px]00[/size][/font][font='times new roman'][size=16px] N/[/size][/font][font='times new roman'][size=16px]m[/size][/font][font='times new roman'][size=16px]。[/size][/font][align=center][img]" style="max-width: 100% max-height: 100% [/img][/align][align=center][font='times new roman']图[/font][font='times new roman']1[/font][font='times new roman'] [/font][font='times new roman'] [/font][font='times new roman']硫脲类物质在[/font][font='times new roman']Sil-P-C12[/font][font='times new roman']色谱柱中的分离谱图[/font][/align][align=center][font='times new roman']色谱条件:流动相,乙腈[/font][font='times new roman']-[/font][font='times new roman']水[/font][font='times new roman']([/font][font='times new roman']97:3[/font][font='times new roman'],[/font][font='times new roman']v/v[/font][font='times new roman']);检测波长,[/font][font='times new roman']214 nm[/font][font='times new roman'];流速,[/font][font='times new roman']1.0 mL/min[/font][/align][align=center][font='times new roman']分析物:[/font][font='times new roman']1. [/font][font='times new roman']二乙基硫脲;[/font][font='times new roman']2. [/font][font='times new roman']亚乙基硫脲;[/font][font='times new roman']3. [/font][font='times new roman']烯丙基硫脲[/font][/align][align=center][font='times new roman']Separation chromatogram of thiocarbamides on the Sil-P-C12 column[/font][/align][align=center][font='times new roman']Chromatographic conditions: mobile phase, ACN-H[/font][font='times new roman'][sub][size=13px]2[/size][/sub][/font][font='times new roman']O (97:3, v/v) detection wavelength, 214 nm flow rate, 1.0 mL/min[/font][/align][align=center][font='times new roman']Analytes: 1. diethyl thiourea 2. ethylene thiourea 3. allylthiourea[/font][/align][align=left][font='times new roman'][size=18px]3.3.4 S[/size][/font][font='times new roman'][size=18px]il[/size][/font][font='times new roman'][size=18px]-P-C12[/size][/font][font='times new roman'][size=18px]色谱柱分离苯酚类化合物[/size][/font][/align][font='times new roman'][size=16px]采用苯酚类化合物进一步考察了[/size][/font][font='times new roman'][size=16px]Sil[/size][/font][font='times new roman'][size=16px]-P-C12[/size][/font][font='times new roman'][size=16px]色谱柱的分离性能。[/size][/font][font='times new roman'][size=16px]图[/size][/font][font='times new roman'][size=16px]2[/size][/font][font='times new roman'][size=16px]为[/size][/font][font='times new roman'][size=16px]对甲酚、对氯苯酚、双酚[/size][/font][font='times new roman'][size=16px]A[/size][/font][font='times new roman'][size=16px]、对乙酰氨基酚和[/size][/font][font='times new roman'][size=16px]2[/size][/font][font='times new roman'][size=16px]-[/size][/font][font='times new roman'][size=16px]羟基苯砜五种苯酚类化合物[/size][/font][font='times new roman'][size=16px]的色谱分离图。以乙腈[/size][/font][font='times new roman'][size=16px]-[/size][/font][font='times new roman'][size=16px]水([/size][/font][font='times new roman'][size=16px]9[/size][/font][font='times new roman'][size=16px]7:3[/size][/font][font='times new roman'][size=16px],[/size][/font][font='times new roman'][size=16px]v/v[/size][/font][font='times new roman'][size=16px])进行洗脱,[/size][/font][font='times new roman'][size=16px]五[/size][/font][font='times new roman'][size=16px]种物质可在[/size][/font][font='times new roman'][size=16px]8[/size][/font][font='times new roman'][size=16px] min[/size][/font][font='times new roman'][size=16px]内达到基线分离,[/size][/font][font='times new roman'][size=16px]分离度分别为[/size][/font][font='times new roman'][size=16px]2.08[/size][/font][font='times new roman'][size=16px]、[/size][/font][font='times new roman'][size=16px]6[/size][/font][font='times new roman'][size=16px].05[/size][/font][font='times new roman'][size=16px]、[/size][/font][font='times new roman'][size=16px]4[/size][/font][font='times new roman'][size=16px].86[/size][/font][font='times new roman'][size=16px]和[/size][/font][font='times new roman'][size=16px]1[/size][/font][font='times new roman'][size=16px]5.63[/size][/font][font='times new roman'][size=16px],最高理论塔板数可达[/size][/font][font='times new roman'][size=16px]7[/size][/font][font='times new roman'][size=16px]7[/size][/font][font='times new roman'][size=16px],900[/size][/font][font='times new roman'][size=16px] N/[/size][/font][font='times new roman'][size=16px]m[/size][/font][font='times new roman'][size=16px],且色谱图峰型良好,[/size][/font][font='times new roman'][size=16px]拖尾因子[/size][/font][font='times new roman'][size=16px]分别为[/size][/font][font='times new roman'][size=16px]1[/size][/font][font='times new roman'][size=16px].09[/size][/font][font='times new roman'][size=16px]、[/size][/font][font='times new roman'][size=16px]1[/size][/font][font='times new roman'][size=16px].07[/size][/font][font='times new roman'][size=16px]、[/size][/font][font='times new roman'][size=16px]0[/size][/font][font='times new roman'][size=16px].94[/size][/font][font='times new roman'][size=16px]、[/size][/font][font='times new roman'][size=16px]0.97[/size][/font][font='times new roman'][size=16px]和[/size][/font][font='times new roman'][size=16px]0[/size][/font][font='times new roman'][size=16px].93[/size][/font][font='times new roman'][size=16px]。[/size][/font][align=center][img]" style="max-width: 100% max-height: 100% [/img][/align][align=center][font='times new roman']图[/font][font='times new roman']2[/font][font='times new roman'] [/font][font='times new roman']苯酚类物质在[/font][font='times new roman']Sil-P-C12[/font][font='times new roman']色谱柱中的分离谱图[/font][/align][align=center][font='times new roman']色谱条件:流动相,乙腈[/font][font='times new roman']-[/font][font='times new roman']水[/font][font='times new roman']([/font][font='times new roman']97:3[/font][font='times new roman'],[/font][font='times new roman']v/v[/font][font='times new roman']);检测波长,[/font][font='times new roman']214 nm[/font][font='times new roman'];流速,[/font][font='times new roman']1.0 mL/min[/font][/align][align=center][font='times new roman']分析物:[/font][font='times new roman']1. 4-[/font][font='times new roman']叔辛基苯酚;[/font][font='times new roman']2. [/font][font='times new roman']对甲酚;[/font][font='times new roman']3. [/font][font='times new roman']双酚[/font][font='times new roman']A[/font][font='times new roman'];[/font][font='times new roman']4. [/font][font='times new roman']对乙酰氨基酚;[/font][font='times new roman']5. 2-[/font][font='times new roman']羟基苯砜[/font][/align][align=center][font='times new roman'] [/font][font='times new roman'] Separation chromatogram of phenols on the Sil-P-C12 column[/font][/align][align=center][font='times new roman']Chromatographic conditions: mobile phase, ACN-H[/font][font='times new roman'][sub][size=13px]2[/size][/sub][/font][font='times new roman']O (97:3, v/v) detection wavelength, 214 nm flow rate, 1.0 mL/min[/font][/align][align=center][font='times new roman']Analytes: 1. 4-[/font][font='times new roman']tert[/font][font='times new roman']-octylphenol 2. [/font][font='times new roman']p[/font][font='times new roman']-cresol 3. bisphenol A 4. acetaminophen 5. 2-hydroxyphenyl sulfone[/font][/align][align=left][font='times new roman'][size=18px]Sil-P-C12[/size][/font][font='times new roman'][size=18px]色谱柱分离磺胺类化合物[/size][/font][/align][font='times new roman'][size=16px]选取磺胺类化合物对[/size][/font][font='times new roman'][size=16px]S[/size][/font][font='times new roman'][size=16px]il[/size][/font][font='times new roman'][size=16px]-P-C12[/size][/font][font='times new roman'][size=16px]色谱柱的分离性能进行进一步评价。图[/size][/font][font='times new roman'][size=16px]3[/size][/font][font='times new roman'][size=16px]为[/size][/font][font='times new roman'][size=16px]磺胺甲恶唑[/size][/font][font='times new roman'][size=16px]、[/size][/font][font='times new roman'][size=16px]磺胺二甲基嘧啶[/size][/font][font='times new roman'][size=16px]、[/size][/font][font='times new roman'][size=16px]乙酰唑胺[/size][/font][font='times new roman'][size=16px]和[/size][/font][font='times new roman'][size=16px]磺胺噻唑[/size][/font][font='times new roman'][size=16px]的分离谱图。[/size][/font][font='times new roman'][size=16px]以乙腈[/size][/font][font='times new roman'][size=16px]-[/size][/font][font='times new roman'][size=16px]水([/size][/font][font='times new roman'][size=16px]97[/size][/font][font='times new roman'][size=16px]:[/size][/font][font='times new roman'][size=16px]3[/size][/font][font='times new roman'][size=16px],[/size][/font][font='times new roman'][size=16px]v/v[/size][/font][font='times new roman'][size=16px])进行洗脱,[/size][/font][font='times new roman'][size=16px]四[/size][/font][font='times new roman'][size=16px]种物质可在[/size][/font][font='times new roman'][size=16px]6[/size][/font][font='times new roman'][size=16px] min[/size][/font][font='times new roman'][size=16px]内达到基线分离,分离度分别为[/size][/font][font='times new roman'][size=16px]3.29[/size][/font][font='times new roman'][size=16px]、[/size][/font][font='times new roman'][size=16px]1[/size][/font][font='times new roman'][size=16px]1[/size][/font][font='times new roman'][size=16px].[/size][/font][font='times new roman'][size=16px]41[/size][/font][font='times new roman'][size=16px]和[/size][/font][font='times new roman'][size=16px]3[/size][/font][font='times new roman'][size=16px].26[/size][/font][font='times new roman'][size=16px],[/size][/font][font='times new roman'][size=16px]理[/size][/font][font='times new roman'][size=16px]论塔板数[/size][/font][font='times new roman'][size=16px]均大于[/size][/font][font='times new roman'][size=16px]5[/size][/font][font='times new roman'][size=16px]0[/size][/font][font='times new roman'][size=16px],[/size][/font][font='times new roman'][size=16px]000 N/[/size][/font][font='times new roman'][size=16px]m[/size][/font][font='times new roman'][size=16px],且色谱图峰型良好,拖尾因子分别为[/size][/font][font='times new roman'][size=16px]1.10[/size][/font][font='times new roman'][size=16px]、[/size][/font][font='times new roman'][size=16px]1[/size][/font][font='times new roman'][size=16px].03[/size][/font][font='times new roman'][size=16px]、[/size][/font][font='times new roman'][size=16px]0[/size][/font][font='times new roman'][size=16px].94[/size][/font][font='times new roman'][size=16px]和[/size][/font][font='times new roman'][size=16px]0[/size][/font][font='times new roman'][size=16px].96[/size][/font][font='times new roman'][size=16px]。[/size][/font][align=center][img]" style="max-width: 100% max-height: 100% [/img][/align][align=center][font='times new roman']图[/font][font='times new roman']3[/font][font='times new roman'] [/font][font='times new roman']磺胺类物质在[/font][font='times new roman']Sil-P-C12[/font][font='times new roman']色谱柱中的分离谱图[/font][/align][align=center][font='times new roman']色谱条件:流动相,乙腈[/font][font='times new roman']-[/font][font='times new roman']水[/font][font='times new roman']([/font][font='times new roman']97:3[/font][font='times new roman'],[/font][font='times new roman']v/v[/font][font='times new roman']);检测波长,[/font][font='times new roman']254 nm[/font][font='times new roman'];流速,[/font][font='times new roman']1.0 mL/min[/font][/align][align=center][font='times new roman']分析物:[/font][font='times new roman']1. [/font][font='times new roman']磺胺甲恶唑;[/font][font='times new roman']2. [/font][font='times new roman']磺胺二甲基嘧啶;[/font][font='times new roman']3. [/font][font='times new roman']乙酰唑胺;[/font][font='times new roman']4. [/font][font='times new roman']磺胺噻唑[/font][/align][align=center][font='times new roman']Fig[/font][font='times new roman'].3[/font][font='times new roman'] Separation chromatogram of sulfonamides on the Sil-P-C12 column[/font][/align][align=center][font='times new roman']Chromatographic conditions: mobile phase, ACN-H[/font][font='times new roman'][sub][size=13px]2[/size][/sub][/font][font='times new roman']O (97:3, v/v) detection wavelength, 254 nm flow rate, 1.0 mL/min[/font][/align][align=center][font='times new roman']Analytes: 1. sulfamethoxazole 2. sulfamethazine 3. acetazolamide [/font][font='times new roman'] [/font][font='times new roman']4. sulfathiazole[/font][/align]
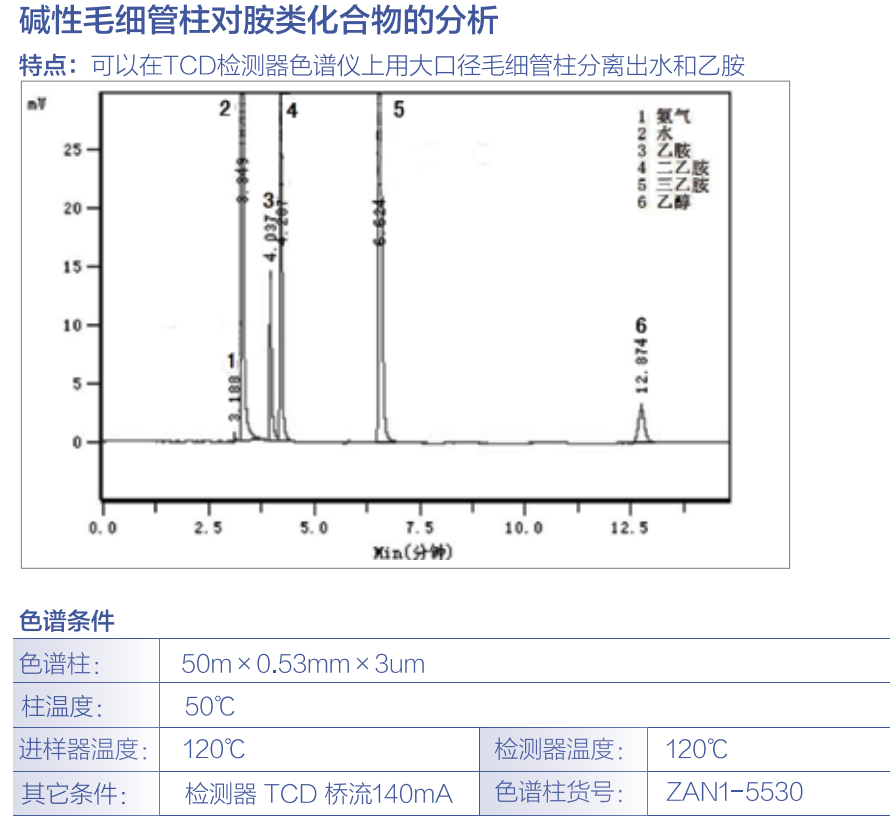
有没有大神做车内空气全谱分析或者车内硫类和胺类化合物测试的??而且是用热脱附GCMS做的??想咨询这方面的问题,私信我,有偿咨询!!!
水质 苯胺类化合物的测定HJ822-2017 净化过程中分别用3次洗脱溶液洗脱,三次的洗脱溶液中分别是哪些物质,苯胺在第几次洗脱液中